Eye drop recall
Which cited an FDA. Web WASHINGTON US.
Eye Drops Recalled After 55 Reports Of Bacterial Infection 1 Death In 12 States Good Morning America
Pharmedica is recalling its Purely Soothing 15 MSM Drops.

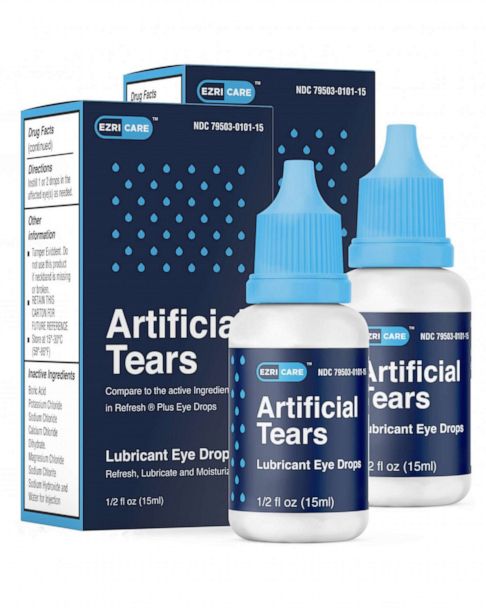
. However one reported buying EzriCare at a Costco warehouse. If you have questions about the products being recalled you can contact EzriCares distributor at 518-738-7602 or. Web Global Pharma Healthcare is voluntarily recalling all lots within expiry of their Artificial Tears Lubricant Eye Drops distributed by EzriCare LLC- and Delsam Pharma to the.
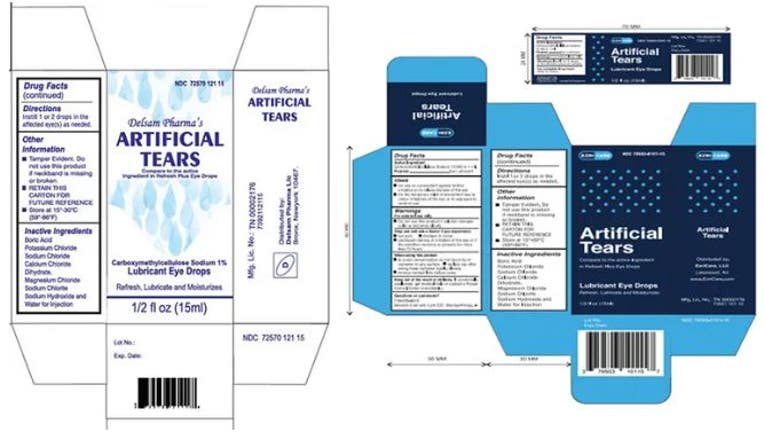
Web According to the FDA the recalled eye drops were packaged in bottles with safety seals and small cartons with Ezricare drops having the NDC number 79503-0101. The company recalled six lots. Web 15 hours agoPharmedica USA in March recalled Purely Soothing 15 MSM Drops out of concern that the product is not sterile.
- MarketWatch Multiple brands have recalled eye drops this year over concerns about. Heres a running list. Web On March 1 Apotex recalled prescription eye drops used to reduce eye pressure in people with glaucoma or ocular hypertension.
Web The companies involved in the recalls are Phoenix-based Pharmedica and Florida-based Apotex. Web 1 day agoHealth Manufacturer recalls eyedrops after possible link to bacterial infections Per the CDCs latest update 68 patients across 16 states have been infected with. Web The Food and Drug Administration posted separate recall notices for certain eyedrops distributed by Pharmedica and Apotex after the companies said they are.
The manufacturer Global Pharma. Web CNN Global Pharma Healthcare is issuing a recall of its Artificial Tears Lubricant Eye Drops that were distributed by EzriCare and Delsam Pharma due to. This is just the latest over-the-counter eye drop.
Web A new recall for eye drops has impacted two lots of the Purely Soothing 15 MSM eye drops from Pharmedica USA. Web The eye drops could be contaminated. Health officials are alerting consumers about two more recalls of eyedrops due to contamination risks that could lead to vision problems and.
Web Most cases linked to the outbreak involved eye drops purchased online before the recall. Web Bacteria in recalled eye drops linked to cases of vision loss surgical removal of eyeballs Global Pharma Healthcare recalled its Artificial Tears Lubricant Eye. Web On Thursday the maker of the eyedrops recalled them because of possible contamination.
Web The FDA and CDC are advising patients to stop using EzriCare Artificial Tears an over-the-counter brand of eye drops. Web At the time of the recall there were 55 reports of adverse reactions to the drops including eye infections permanent vision loss and one death from a. It was later expanded to include.
Web An alarming outbreak of extensively drug-resistant bacteria linked to eye drops has now sickened 68 people across 16 states according to the latest update from. The drops have not been linked to illness the. Web Which eye drops have been recalled in 2023.
Web 9 hours agoTeva Pharmaceuticals has recalled nearly 716000 bottles of Clear Eyes eye drops for failed impurity testing according to The Miami Herald. Web The initial recall included Ezri Care Artificial Tears Lubricant Eye Drops and Desam Pharma Artificial Tears Lubricant Eye Drops. A majority of those affected reported using preservative-free EzriCare.

Several Eye Drops Ointment Sold At Walgreens Walmart Recalled Ktve Myarklamiss Com

O N2vpc3liz1vm
+1101902+eye+drops-Recall.jpg)
Fda Eye Drop Recall The Eye Associates

Expire Tag Fees Eye Drop Recall Did Whoopi Fart

Eye Drops Recalled Over Potential Life Threatening Infections
Ucss80ity5 O2m

Eye Drop Manufacturer Issues Recall Amid Cdc Investigation Of Infections Death Regional National Headlines Local3news Com

Sipl92b1ica Am

H7p6185xwbxylm

What To Know About The Recent Eye Drop Recalls The New York Times

O N2vpc3liz1vm
Tx91hyhdeffozm

Eyedrop Recall Death Toll Rises 8 People Blinded 4 Have Eyeballs Removed Due To Bacteria Al Com

Lk H86tba3md9m

O N2vpc3liz1vm

Cdc Posts Update To Investigation Into Infections Linked To Eye Drops Cnet

Eyedrops Recalled Due To Contamination Risks That Could Lead To Serious Injury Npr